
Your development partner for
medical device software engineering
We help you evolve legacy medical device software
in a structured way — with sound engineering,
aligned with your regulatory processes, and focused
on reliable evidence and stable releases.

Are you looking for a partner who can reliably advance your device software in a regulated environment? We support the development of software and applications for medical devices and medical technology systems.
In medical technology, “somehow working” is not enough. Changes must be implemented in a traceable way, tests must be reliable, and results must remain properly documented. At the same time, many teams face a specific challenge: the software runs reliably on the device, but every extension — whether new features, additional interfaces, or adaptations — becomes increasingly complex and difficult to estimate.
As an external development partner, we work within your existing processes. Responsibility for regulatory compliance, the quality management system, and approval remains with you. We provide code, tests, and supporting evidence in a way that integrates into your framework. This typically includes development artifacts aligned with IEC 62304, documented changes linked to risk, and traceability that supports your audits and technical documentation.
Qwasaa helps you advance existing device software in a structured way — within your current system, typically in C++/Qt or C#/.NET, and aligned with your regulatory requirements.
We structure implementation and coordination so that decisions remain transparent and quality assurance is not treated as a downstream step, but as an integral part of development.
The result:
Changes become more predictable, evidence more robust, and releases more stable — so your software performs reliably in daily device operation.

Specialized device software for regulated environments
Software in medical technology performs critical functions: it controls device features, processes measurement data, and presents information in a way that is easy to understand and safe to use in day-to-day operations. Unlike in many other embedded domains, technical decisions here have a direct impact on risk assessment, approval documentation, and service processes — every change has both a technical and a regulatory dimension.
Typical challenges include:
- Changes to the code affect risk analysis, verification, and the technical documentation
- Dependencies between software components, hardware, and the operating environment are not always fully documented
- New requirements, for example from post-market surveillance or updated standards, must be integrated into legacy structures
- Tests and supporting evidence must remain robust, even when parts of the architecture are further developed
In this environment, software engineering means implementing changes in a way that is technically sound and remains traceable for your regulatory documentation. We work with the artifacts defined by your quality management system and deliver them in a form that fits into your processes.
How Qwasaa supports you
Our software development services for medical devices.
Targeted enhancement and modernization of your device software — aligned with your existing architecture and processes.
Changes are integrated in a traceable manner and coordinated cleanly.
Test strategy, test planning, and automation help identify issues early.
Connecting equipment, data sources, and existing systems to improve analysis and create more stable workflows.
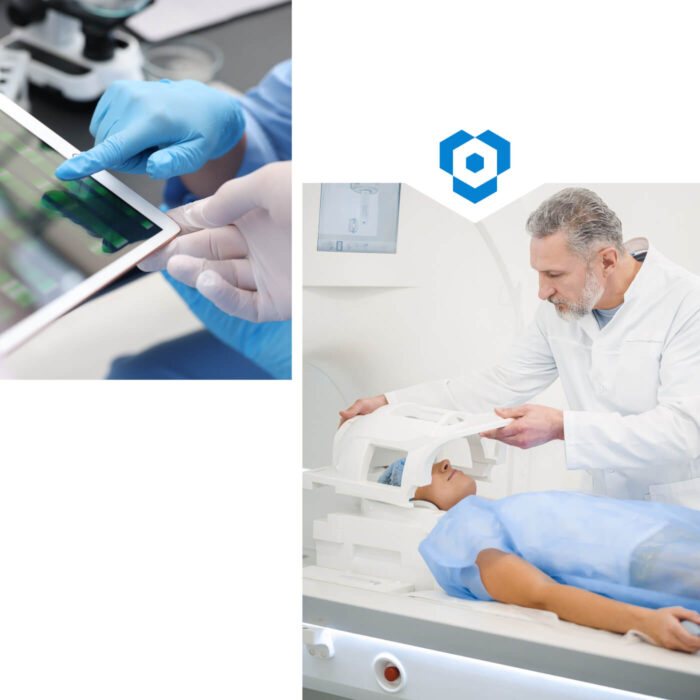
Typical application areas for software in medical technology
- Control and monitoring of medical devices
- Processing and visualization of measurement data
- User guidance for safe day-to-day operation
- Integration of devices into higher-level systems
Technologies and environment
We work in environments where such systems are typically developed:
- C++ / Qt
- C# / .NET
- Device connectivity and technical interfaces
- UI development focused on clarity and safe operation
- Long-lived codebases and legacy architectures
When device software extends beyond the device itself
Device data in service and backend processes
In many projects, the value of device software does not end at the device itself. Device-related information — such as statuses, usage data, service cases, or results — also needs to be available outside the device: for customer care, technical documentation, post-market surveillance, or data analysis.
This is typically where additional applications are needed to capture device data in a structured way, make it available, and integrate it into existing service and backend processes. Depending on their purpose and data flow, such applications may also be subject to regulatory requirements and must therefore be developed in a traceable manner.
An example from our project work:
For one manufacturer, we implemented a service portal that captures device data in a structured way and makes it usable for internal follow-up processes. The application integrates into the existing backend landscape and is designed so that changes can be implemented in a controlled and documented way. It was built in PHP/Symfony and embedded into the customer’s existing system environment.
Typical aspects include:
- structured capture and provision of device data
- integration into existing backend and service processes
- user-friendly interfaces for internal or external users
- clear separation between device software and surrounding systems
The result:
- more stable releases
- traceable tests and supporting evidence
- predictable changes
- better maintainability
- software that works reliably in real-world use
Let’s discuss your system and current situation.
We will tell you openly where we can support you and what the most sensible next step is.
Let’s get started
We look forward to your inquiry and will get back to you within one business day.
By clicking the submit button, you confirm that you agree to our privacy policy.
You can find us here
Our home is the 13th floor of the JenTower in the heart of the beautiful city of Jena on the Saale.
Feel free to stop by – we look forward to seeing you.
show on map

